A modular system is meant to contain your entire process. It can be a water-for-injection (WFI) distribution skid or a custom process for your operation. When a manufacturer takes responsibility of your equipment requirements at their facility, you can minimize your risks while maximizing your speed to market. Below are examples of how a skid-mounted system could benefit your next project.
Reduced Time to Start-Up
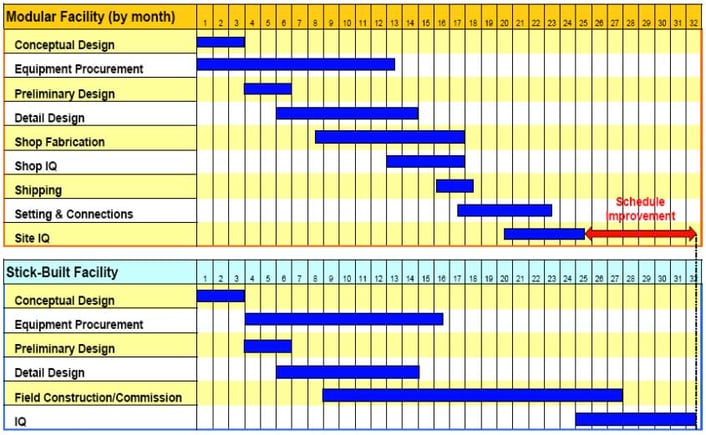
Modular skids are ideal for your tight schedule as they drastically reduce the system set-up time by eliminating the need for multiple vendors or the stick-built process. Sole-sourcing from one vendor reduces your time and labor poured into evaluating bids, purchase orders, specs, change orders, and documentation for multiple pieces of equipment. When a modular system is implemented, design, fabrication, and testing of your equipment will be simultaneous with the construction of your building, resulting in a vastly reduced set-up time. Your equipment’s completion can be scheduled to fit in your ideal time frame so your facility can remain open for installation for the appropriate amount of time eliminating or reducing downtime.
The International Society of Pharmaceutical Engineers (ISPE) performed a case study on two units in an oral dosage facility in Puerto Rico to see how having a modular facility affects schedule. The modular facility was completed faster by more than six months over the stick-built facility.
Reduced Labor Risk
Modular skids are also ideal for areas lacking in local resources as sourcing a skid can make up for the lack of welders or grinders in your area. Bringing in these craftsmen dramatically increases project pricing through travel and housing expenses, while having manufacturing experts working at a controlled site takes away the hassle of searching for qualified craftsmen.
Maintenance of Higher Quality Standards
Fabrication and validation of your equipment should be performed in an orderly and controlled shop environment. Your manufacturer’s facility should be equipped with final assembly and test areas that are climate-controlled and isolated from welding and grinding areas. Your supplier will be held accountable for managing your project’s multiple needs resulting in cohesive quality and reliability throughout all parts of your equipment. You should expect your equipment to be fully tested in a proper environment before shipment, preventing any issues on-site.
Improved Documentation
Modular skid documentation will come in binders with consistent documentation, forms, and shipment times. A modular skid turnover package (TOP) is easier to understand and keep track of compared to multiple TOPs from various vendors. Documentation will be formatted in the same template throughout, and you don’t need to worry about varying arrival times and physical forms. Individual equipment within the skid will have its own TOP. These TOPs can then reference one another, providing uniformity and clarity. Using multiple vendors means multiple TOPs which could be disconnected and confusing.
Modular skids have many advantages that can help your project run more efficiently. Contact your pharmaceutical equipment manufacturer to see how they can meet your process requirements with a skid-mounted system.
Looking to design your own skid? Check out our Tank Volume Calculator and 3D Models to get started today!


